UL Launches Medical IoT Battery SOH Transparency Certification Pilot
author
UL Solutions announced the launch of its ‘Medical IoT Battery State-of-Health Transparency’ certification pilot program on May 5, 2026 — a development with direct implications for manufacturers of continuous glucose monitors (CGMs), portable ECG devices, and related battery-powered medical IoT hardware. This initiative signals an emerging expectation for real-time battery health visibility in clinical-grade wearable and point-of-care devices, making it relevant for battery module suppliers, medical device OEMs, and regulatory affairs teams operating across U.S. and global markets.
Event Overview
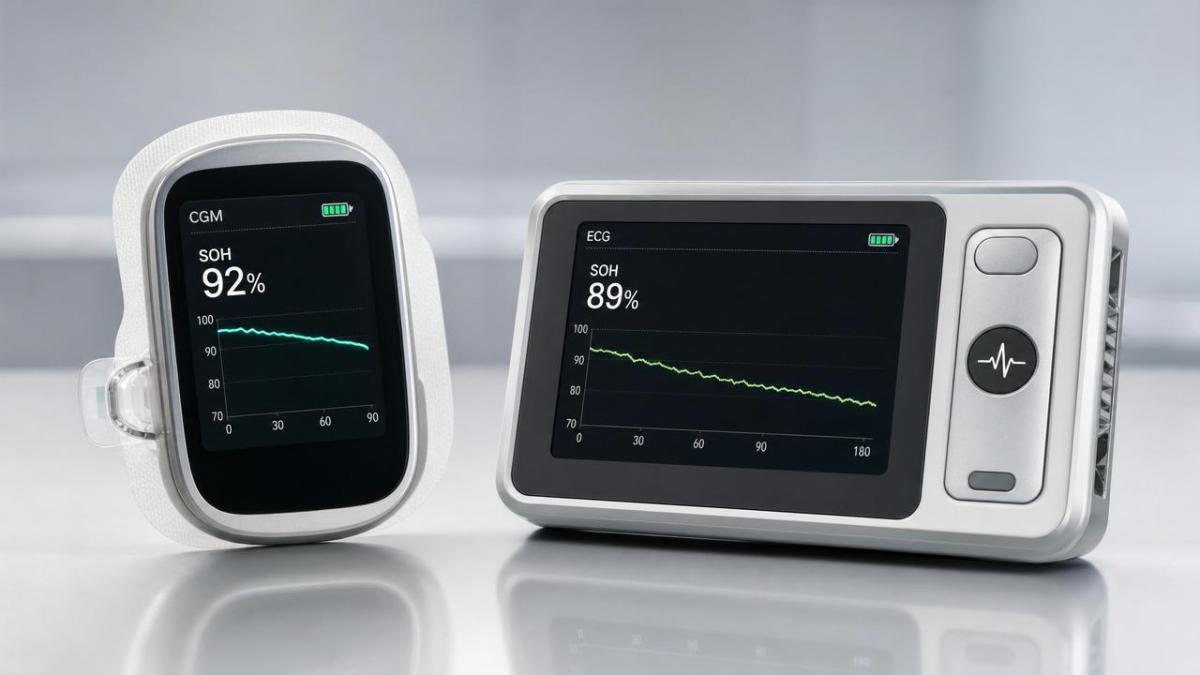
On May 5, 2026, UL Solutions initiated a pilot program titled ‘Medical IoT Battery State-of-Health Transparency’. The program requires lithium-ion batteries used in medical IoT devices — including continuous glucose monitors (CGMs) and portable electrocardiogram (ECG) instruments — to display real-time State-of-Health (SOH) values and degradation curves within the device’s user interface or companion mobile application. Compliance is verified under an addendum to UL 2054. Three battery module suppliers based in China have been selected for the initial pilot phase.
Industries Affected by Segment
Battery Module Suppliers (OEM/ODM Level)
Suppliers providing integrated lithium-ion battery packs to medical device makers are directly affected. The requirement to embed SOH estimation algorithms, telemetry interfaces, and UI-ready data formatting introduces new firmware, validation, and documentation obligations beyond standard safety certification.
Medical Device OEMs (CGM, Portable ECG, Remote Monitoring)
OEMs integrating third-party battery modules must now assess whether their current hardware and software architectures support real-time SOH rendering — including display logic, data parsing, and user-facing visualizations. This may trigger updates to firmware architecture, UI design specs, and human factors validation protocols.
Regulatory & Compliance Service Providers
Firms supporting medical device certification must expand scope coverage to include SOH transparency as a functional safety and usability element — not only under UL 2054 but potentially as input to IEC 62304 (software lifecycle) and IEC 62366-1 (usability engineering) assessments.
What Relevant Enterprises or Practitioners Should Monitor and Do Now
Track official scope definition and timeline extension
Analysis shows the pilot remains limited in scope and duration; stakeholders should monitor UL Solutions’ public updates for formal expansion plans, inclusion of additional device categories (e.g., insulin pumps, pulse oximeters), and potential alignment with FDA guidance or EU MDR post-market surveillance expectations.
Assess SOH implementation feasibility per product class
Observably, real-time SOH display imposes distinct technical constraints depending on device class: low-power CGMs may require ultra-low-overhead estimation methods, while higher-compute portable ECGs could leverage onboard analytics. Companies should map current battery management IC capabilities and firmware readiness against UL’s upcoming verification criteria.
Distinguish between pilot requirements and enforceable standards
Current more appropriately understood as a signal rather than a mandate: no regulatory body has adopted SOH transparency as a compliance requirement yet. Enterprises should avoid premature full-scale redesigns but initiate internal gap assessments — particularly around data traceability, algorithm validation, and UI consistency with ISO 14971 risk management outputs.
Engage early with battery module suppliers on data interface specifications
From industry perspective, interoperability between device firmware and battery module telemetry is foundational. Companies should proactively align on communication protocols (e.g., SMBus, custom UART framing), SOH calculation methodology disclosure, and calibration update mechanisms — especially where multiple suppliers serve the same OEM.
Editorial Perspective / Industry Observation
This pilot is best interpreted as an early-stage market-shaping initiative rather than an imminent regulatory threshold. Analysis shows UL is testing technical feasibility, stakeholder readiness, and clinical utility perception — not enforcing compliance. Observably, it reflects growing attention to battery-related reliability in long-term remote monitoring use cases, where unexpected capacity loss can impact clinical decision-making continuity. From industry angle, the move underscores a broader shift: battery performance is evolving from a passive safety parameter into an active, user-facing functional attribute — one that intersects electrical safety, software validation, and human factors engineering.
It is not yet a standard, nor does it carry legal enforcement weight. However, its timing — amid increasing FDA focus on AI/ML-enabled device transparency and EU MDR emphasis on lifetime performance data — suggests it may inform future guidance or harmonized test methods. Continued observation is warranted, particularly for companies supplying into U.S.-regulated markets or pursuing dual-market (U.S./EU) approvals.
Conclusion
This pilot represents an early indicator of converging expectations across safety certification, clinical usability, and battery intelligence in medical IoT. It does not introduce new regulation, but it does redefine the baseline for technical dialogue between battery suppliers and medical device developers. Currently, it is more accurately understood as a readiness probe — one that invites structured evaluation, not immediate compliance overhaul.
Source Attribution
Main source: UL Solutions official announcement (May 5, 2026).
Points requiring ongoing observation: formal publication of UL 2054 addendum language, expansion beyond initial three Chinese suppliers, and any linkage to FDA or EU notified body guidance.
Protocol_Architect
Dr. Thorne is a leading architect in IoT mesh protocols with 15+ years at NexusHome Intelligence. His research specializes in high-availability systems and sub-GHz propagation modeling.
Related Recommendations
- Dr. Sophia Carter (Medical IoT Specialist)UL Launches Medical IoT Battery SOH Transparency Certification Pilot
- Dr. Sophia Carter (Medical IoT Specialist)UL Launches Medical IoT Battery SOH Transparency Pilot
- Dr. Sophia Carter (Medical IoT Specialist)UL 2900-2-2:2026 Enforces AI Robustness Testing for Medical IoT in US Market
- Dr. Sophia Carter (Medical IoT Specialist)UL 2900-2-2 Updated: AI Models in Medical IoT Must Pass Adversarial Robustness Testing
- Dr. Sophia Carter (Medical IoT Specialist)UL 2900-2-2 Rev.4: AI Robustness Testing Mandatory for Medical IoT
- Dr. Sophia Carter (Medical IoT Specialist)UL 2900-2-2 Ed.4 Adds Adversarial Robustness Testing for Medical IoT AI
- Dr. Sophia Carter (Medical IoT Specialist)Medical IoT Deployment Challenges in Off-Grid Facilities
- Dr. Sophia Carter (Medical IoT Specialist)Why Medical IoT Depends on Reliable Renewable Power
- Dr. Sophia Carter (Medical IoT Specialist)Medical IoT Power Backup Risks in Remote Energy Sites
- Dr. Sophia Carter (Medical IoT Specialist)UL 2900-2-2 Ed.3: AI Robustness Testing Mandatory for Medical IoT
Analyst
